Multiple Guess!!-Select the best answer for each question or
problem.
|
|
|
1.
|
How is 0.00069 written in
scientific notation?
a. | 69 ´ 10–5 | b. | 6.9 ´ 104 | c. | 0.69 ´
10–3 | d. | 6.9 ´
10–4 |
|
|
|
2.
|
Which of the following
conversion factors would you use to change 18 kilometers to meters?
a. | 1000 m/1
km | c. | 100 m/1
km | b. | 1 km/1000
m | d. | 1 km/100 m |
|
|
|
3.
|
Timers at a swim meet used four
different clocks to time an event. Which recorded time is the most precise?
a. | 55 s | b. | 55.2 s | c. | 55.25 s | d. | 55.254 s |
|
|
|
4.
|
A beaker contains 0.32 L of
water. What is the volume of this water in milliliters?
a. | 320 mL | b. | 3.2 mL | c. | 32 mL | d. | 0.32
mL |
|
|
|
5.
|
The radius of Earth is 6 370
000 m. Express this measurement in km in scientific notation with the correct number of significant
digits.
|
|
|
6.
|
Three values were obtained for
the mass of a metal bar: 8.83 g; 8.84 g; 8.82 g. The known mass is 10.68 g. The values
are
a. | accurate. | c. | both accurate and precise. | b. | precise. | d. | neither accurate nor precise. |
|
|
|
7.
|
Calculate the following, and
express the answer in scientific notation with the correct number of significant figures: 21.4 + 15 +
17.17 + 4.003
|
|
|
8.
|
Calculate the following, and
express the answer in scientific notation with the correct number of significant figures: 10.5
´ 8.8 ´ 3.14
a. | 2900 | b. | 290.136 | c. | 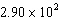 | d. | 290 |
|
|
|
|
|
|
9.
|
Which best describes this
graphical relationship?
a. | direct | b. | indirect | c. | inverse | d. | impossible |
|
|
|
|
|
|
10.
|
Which best describes this
graphical relationship?
a. | impossible | b. | inverse | c. | indirect | d. | direct |
|
|
|
11.
|
Which is the correct
measurement for location marked by the arrow?
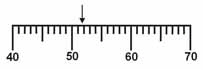
|
|
|
12.
|
The accepted value is 29.35.
Which correctly describes this student’s experimental data?
| Trial | Measurement | | 1 | 29.48 | | 2 | 28.97 | | 3 | 29.27 | | |
a. | accurate but not
precise | c. | both accurate and
precise | b. | precise but not accurate | d. | neither accurate nor precise |
|
|
|
13.
|
The accepted value is 1.43.
Which correctly describes this student’s experimental data?
| Trial | Measurement | | 1 | 1.29 | | 2 | 1.93 | | 3 | 0.88 | | |
a. | accurate but not
precise | c. | both accurate and
precise | b. | precise but not accurate | d. | neither accurate nor precise |
|
|
|
14.
|
Which value has only 4
significant digits?
a. | 6.930 | b. | 0.0450 | c. | 8450 | d. | 0.392 |
|
|
|
15.
|
Which is the sum of these
values, to the appropriate number of significant digits?
17.358 + 3.502 + 20.14 =
a. | 41 | b. | 41.00 | c. | 41.000 | d. | 41.0 |
|
|
|
16.
|
Which is the product of these
numbers, to the appropriate number of significant digits?
56.2 ´ 9.2057 =
a. | 517 | b. | 517.4 | c. | 517.36 | d. | 517.00 |
|
|
|
17.
|
How many significant digits are
in the value 530,405,000?
|
|
|
18.
|
How many significant digits are
in the value 0.0050340?
|
|
|
19.
|
How many minutes are in 1
week?
a. | 168 min | b. | 1440 min | c. | 10 080 min | d. | 100 800
min |
|
|
|
20.
|
If repeated measurements agree
closely but differ widely from the accepted value, these measurements are
a. | neither precise nor
accurate. | b. | accurate, but not precise. | c. | both precise and accurate. | d. | precise, but not accurate. |
|
|
|
21.
|
The measurement 0.035550 g
rounded off to two significant figures would be
a. | 0.03 g. | b. | 0.35 g. | c. | 0.036 g. | d. | 3.5 ´ 102
g. |
|
|
|
22.
|
Which of these measurements has
been expressed to three significant figures?
a. | 0.052 g | b. | 0.202 g | c. | 3.065 g | d. | 500
g |
|
|
|
23.
|
Which of these measurements has
been expressed to four significant figures?
a. | 0.0020
mm | b. | 0.0402
mm | c. | 30.00
mm | d. | 402.10
mm |
|
|
|
24.
|
When 64.4 is divided by 2.00,
the correct number of significant figures in the result is
|
|
|
25.
|
How is the measurement 0.000
065 cm written in scientific notation?
a. | 65 ´ 10–6 cm | c. | 6.5 ´ 10–6 cm | b. | 6.5 ´ 10–5
cm | d. | 6.5 ´ 10–4 cm |
|
|
|
26.
|
The speed of light is 300 000
km/s. In scientific notation, this speed is written to one significant figure as
a. | 3 ´ 105 km/s. | c. | 3. ´ 106 km/s. | b. | 3.0 ´ 105
km/s. | d. | 3.0 ´ 106 km/s. |
|
|
|
27.
|
The closeness of a measurement
to its true value is a measure of its ____.
a. | precision | b. | accuracy | c. | reproducibility | d. | usefulness |
|
|
|
28.
|
In the measurement 0.503 L,
which digit is the estimated digit?
a. | 5 | c. | 3 | b. | the 0 immediately to the left of the
3 | d. | the 0 to the left of the decimal
point |
|
|
|
29.
|
What is the temperature of
absolute zero measured in  C? C?
a. | –373 C C | b. | –273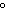 C C | c. | –173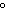 C C | d. | –73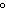 C C |
|
|
|
30.
|
If the temperature changes by
100 K, by how much does it change in 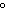 C? C?
|